THP1-Dual™ KO-MAVS Cells
-
Cat.code:
thpd-komavs
- Documents
ABOUT
KO-MAVS dual reporter monocytes for RLR pathway studies
InvivoGen offers a new series of THP1-Dual™ cell lines, derived from the human THP-1 monocytic cell line, and specifically designed for assessing the role of RIG-I, MDA5, and MAVS in the cytosolic RNA sensing pathways.
THP1-Dual™ KO-MAVS cells express two inducible reporter genes, allowing the concomitant study of the IRF and NF-κB pathways, by monitoring the Lucia luciferase and SEAP (secreted embryonic alkaline phosphatase) activities, respectively. In addition, these cells feature stable knockout of the MAVS (Mitochondrial antiviral-signaling protein) gene.
RIG-I and MDA5 are two distinct sensors of viral double-stranded RNA (dsRNA), a replication intermediate for RNA viruses [1-3]. Upon recognition of dsRNA, RIG-I and MDA5 are recruited by the MAVS adaptor to the outer membrane of the mitochondria leading to the activation of several transcription factors, including interferon regulatory factors (IRFs) and NF-κB [3].
Key features
- Verified knockout of MAVS genes (PCR, DNA sequencing, Western blot, and functional assays)
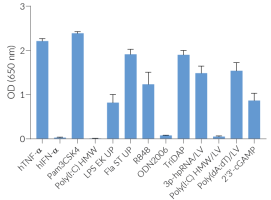
- Functionally validated with a selection of PRR ligands and cytokines
- Readily assessable Lucia luciferase and SEAP reporter activities
- The stability for 20 passages, following thawing, has been verified
- Guaranteed mycoplasma-free
Application
- Defining the role of MAVS in cytosolic RNA sensing pathways
- Highlighting possible overlap between RIG-I, MDA5, MAVS, and STING signaling functions
- Developing novel specific inhibitors of the RLR signaling pathway
References
1. Gebhardt A. et al., 2017. Discrimination of Self and Non-Self Ribonucleic Acids. Journal of Interferon & Cytokine Research 37: 184-97.
2. Pichlmair A. et al., 2006. RIG-I mediated antiviral responses to single-stranded RNA bearing 5’-phosphates. Science 314:997-1001.
3. Kawai T. et al., 2005. IPS-1, an adaptor triggering RIG-I- and Mda5-mediated type I interferon induction. Nat Immunol. 6(10):981-988.
Disclaimer: These cells are for internal research use only and are covered by a Limited Use License (See Terms and Conditions). Additional rights may be available.
SPECIFICATIONS
Specifications
Screening of PRR ligands and cytokines
Complete RPMI 1640 (see TDS)
Verified using PlasmotestTM
Each lot is functionally tested and validated.
CONTENTS
Contents
-
Product:THP1-Dual™ KO-MAVS Cells
-
Cat code:thpd-komavs
-
Quantity:3-7 x 10^6 cells
- 1 ml of Blasticidin (10 mg/ml)
- 1 ml of Zeocin® (100 mg/ml)
- 1 ml of Normocin™ (50 mg/ml)
- 1 tube of QUANTI-Luc™ 4 Reagent (sufficient to prepare 25 ml)
- 1 ml of QB reagent and 1 ml of QB buffer (sufficient to prepare 100 ml of QUANTI-Blue™ Solution)
Shipping & Storage
- Shipping method: Dry ice
- Liquid nitrogen vapor
- Upon receipt, store immediately in liquid nitrogen vapor. Do not store cell vials at -80°C.
Storage:
Caution:
Details
MAVS (mitochondrial antiviral-signaling protein, also known as IPS‑1, CARDIF, VISA)
MAVS is an adaptor protein that plays a critical role in the immune response to viral infection. The innate immune system senses intracellular double-stranded RNA (dsRNA), a replication intermediate for RNA viruses, through two RNA helicases: retinoic acid-inducible gene-I (RIG-I) and melanoma differentiation-association gene 5 (MDA5). Upon recognition of dsRNA, RIG-I and MDA5 are recruited by MAVS to the outer membrane of the mitochondria leading to the activation of several transcription factors including interferon-regulatory factor 3 (IRF3), IRF7, and NF-κB. IRFs and NF-κB regulate the expression of type I interferons (IFNs) and pro-inflammatory cytokines, respectively [1, 2].
RIG-I (retinoic-acid-inducible protein 1, also known as Ddx58)
RIG-I is a cytoplasmic RNA helicase that is critical for host antiviral responses. It senses double-stranded RNA (dsRNA), a replication intermediate for RNA viruses, leading to the production of type I interferons (IFNs) [1]. RIG-I binds specifically to short dsRNAs that have blunt ends and a 5’-triphosphate (5’-ppp) moiety, facilitating discrimination between host and viral dsRNA [3].
MDA5 (melanoma-differentiation-associated gene 5, also known as Ifih1 or Helicard)
MDA5 is a cytoplasmic RNA helicase that plays an important role in antiviral response. It senses long double-stranded RNA (dsRNA), a replication intermediate for RNA viruses, leading to the production of type I interferons (IFNs) [1]. MDA-5 and the related RNA helicase RIG-I recognize a complementary set of cytosolic viral dsRNA. Transfected Poly(I:C), a synthetic analog of viral dsRNA, is recognized by both MDA-5 and RIG-I.
1. Gebhardt A. et al., 2017. Discrimination of Self and Non-Self Ribonucleic Acids. Journal of Interferon & Cytokine Research 37: 184-97.
2. Kawai T. et al., 2005. IPS-1, an adaptor triggering RIG-I- and Mda5-mediated type I interferon induction. Nat Immunol. 6(10):981-988.
3. Pichlmair A. et al., 2006. RIG-I mediated antiviral responses to single-stranded RNA bearing 5’-phosphates. Science 314:997-1001.
DOCUMENTS
Documents
Technical Data Sheet
Validation Data Sheet
Safety Data Sheet
Certificate of analysis
Need a CoA ?