Conjugatable PRR ligands are synthetic PAMPs that have been chemically modified to allow their attachment to a peptide/protein with a chemical linker. Their assembly generates a bioconjugate. Examples of promising therapeutic bioconjugates include antibody-drug conjugates (ADCs, also known as ISACs) and antigen-adjuvant conjugates (AACs). InvivoGen offers conjugatable ligands for PRRs that are expressed by dendritic cells and other antigen-presenting cells. The targeting of TLR7 or STING is of particular interest as it triggers the production of pro-inflammatory cytokines potentiating immune responses in cancer or pathogen-driven disease models.
The ligand
The ligand is a synthetic molecule (PAMP) that is recognized by a specific PRR and triggers (agonist) or blocks (antagonist) inflammatory responses. It features a reaction handle (e.g. free amine, carboxylic acid, azido group) allowing controlled reaction with the linker.
The peptide/protein of interest (POI)
POIs (e.g. monoclonal antibodies, antigenic peptides/proteins) must feature solvent-accessible amino-acidic conjugation sites, cysteines and lysines being most commonly utilized.
The linker
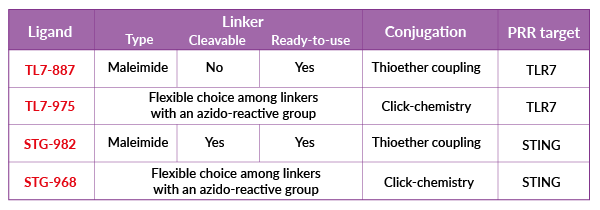
The linker is a chemical moiety that covalently bridges the ligand and POI. Linkers can be categorized as non-cleavable or cleavable (for a localized release of the ligand). They can also feature a single or two functional groups. Linkers featuring a single functional group react selectively with amino-acidic residues (e.g. cysteine or lysine) on the POI. Linkers featuring two functional groups react selectively with amino-acidic residues (e.g. cysteine or lysine) on the POI and azido groups on the ligand.
Conjugation strategies
Commonly described chemical reactions used for bioconjugation include:
- Thioether coupling: A reaction between cysteine-free thiols or sulfur-modified lysines on the POI and activated thio-reactive maleimide handles on the linker (or linker+ligand).
- Amide coupling: A reaction between lysine-free amines on the POI and activated amine-reactive carboxylic acid handles on the linker (or linker+ligand).
- Click-chemistry coupling: A spontaneous reaction between an azido group on the ligand and an azido-reactive group on the linker.
Successful bioconjugation requires (1) designing of the linker, (2) modification of the ligand to react with the linker, (3) treatment of the POI to expose amino-acidic conjugation sites.
![]() Learn more about bioconjugation
Learn more about bioconjugation
To facilitate your research on ADCs and vaccination, InvivoGen has developed two classes of conjugatable ligands allowing:
- Simple thioether conjugation: ready-to-use ligands featuring a thio-reactive maleimide, bypassing the need for linker design and ligand modification.
- Flexible conjugation: ligands featuring an azido group, offering a flexible choice among commercially available linkers for click chemistry conjugation.