Brefeldin A
-
Cat.code:
inh-bfa
- Documents
ABOUT
Inhibitor of STING trafficking
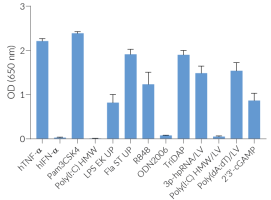
Brefeldin A (BFA) is a fungal macrocyclic lactone and a potent, reversible inhibitor of intracellular vesicle formation and protein trafficking between the endoplasmic reticulum (ER) and the Golgi apparatus [1]. BFA and its analogs are promising inhibitors in drug development due to a number of key features such as apoptosis‑inducing properties as well as antitumor, antifungal, and antiviral effects [2]. Interestingly, despite effectively impairing NLRP3 inflammasome activation, BFA does not block the release of IL-1β, for which the secretion mechanism remains elusive [3].
Mode of action:
The blocking of intracellular vesicle movement in BFA-treated cells causes the rapid accumulation of proteins in the ER, thereby disrupting the trafficking of many proteins. Hence, BFA is commonly utilized as an inhibitor of protein secretion in cellular assays.
BFA also effectively inhibits the secretion of cytokines by blocking the trafficking of upstream signaling proteins. BFA inhibits type I interferon (IFN) production by blocking the dissociation of activated STING (stimulator of interferon genes) from the ER. This prevents the movement of STING to the ER-Golgi intermediate compartment (ERGIC), where it activates the TBK1-IRF3 signaling axis, and ultimately triggers the expression of IFNs [4].
Key Features of Brefeldin A:
- Brefeldin A is a potent inhibitor of protein trafficking from the ER to the Golgi apparatus.
- Brefeldin A inhibits the translocation of STING to the ERGIC, thereby blocking STING-induced type I IFN production.
- InvitroFit™: each lot of Brefeldin A is highly pure (≥95%) and functionally tested.
References:
1. Chardin, P. & McCormick, F. 1999. Brefeldin A: the advantage of being uncompetitive. Cell 97, 153-155.
2. Paek, S. M. 2018. Recent Synthesis and Discovery of Brefeldin A Analogs. Mar Drugs 16.
3. Hong, S. et al., 2019. Brefeldin A-sensitive ER-Golgi vesicle trafficking contributes to NLRP3-dependent caspase-1 activation. FASEB J 33, 4547-4558.
4. Dobbs, N. et al., 2015. STING Activation by Translocation from the ER Is Associated with Infection and Autoinflammatory Disease. Cell Host Microbe 18, 157-168.
All products are for research use only, and not for human or veterinary use.
InvitroFit™
InvitroFit™ is a high-quality standard specifically adapted for in vitro studies of inhibitors. InvitroFit™ products are highly pure (≥95%) and guaranteed free of bacterial contamination, as confirmed using HEK Blue™ TLR2 and HEK Blue™ TLR4 cellular assays. Each lot is rigorously tested for functional activity using validated (or proprietary) cellular models. This grade ensures reliability and reproducibility for your research applications.
SPECIFICATIONS
Specifications
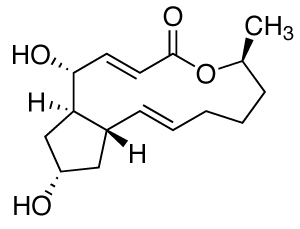
C16H24O4
10 mg/ml in DMSO
1 - 10 μM for cell culture assays
Negative (tested using EndotoxDetect™ assay)
In vitro cellular assays
Each lot is functionally tested and validated using cellular assays.
CONTENTS
Contents
-
Product:Brefeldin A
-
Cat code:inh-bfa
-
Quantity:10 mg
Brefeldin A is provided as an evaporated translucent film.
Shipping & Storage
- Shipping method: Room temperature
- -20 °C
- Avoid repeated freeze-thaw cycles
Storage:
Caution:
Details
Chemical structure of Brefeldin A:
DOCUMENTS
Documents
Technical Data Sheet
Safety Data Sheet
Validation Data Sheet
Certificate of analysis
Need a CoA ?