OxPAPC
-
Cat.code:
tlrl-oxp1
- Documents
ABOUT
TLR2 and TLR4 inhibitor
Oxidized PAPC (oxPAPC) is a bioactive principal component of minimally modified low-density lipoprotein (MM-LDL). OxPAPC inhibits signaling via Toll-like receptor 2 (TLR2) and TLR4 [1]. TLR2 and TLR4 are pattern recognition receptors that recognize bacterial cell wall components such as lipopeptides and lipopolysaccharide (LPS).
Mode of action:
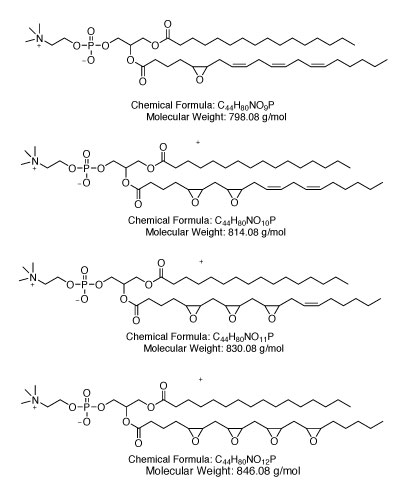
OxPAPC is generated by the oxidation of 1-palmitoyl-2-arachidonyl-sn-glycero- 3-phosphorylcholine (PAPC), a major component of mammalian cell membranes that becomes oxidized by reactive oxygen species (ROS) released from dead or dying cells [2].
The oxidation of PAPC results in a mixture of oxidized phospholipids containing either fragmented or full-length oxygenated sn-2 residues. Both fragmented and full‑length oxygenated species can modulate inflammatory responses. Oxidized phospholipids play important roles in multiple physiological and pathophysiological conditions [2].
OxPAPC has been shown to inhibit the signaling induced by bacterial lipopeptides and lipopolysaccharide (LPS). It acts by competing with LPS-binding protein (LBP), CD14 and, myeloid differentiation protein 2 (MD-2), the accessory proteins that interact with bacterial lipids, thus blocking the signaling of TLR2 and TLR4 [1, 3]. Interestingly, it has been suggested that in animal models OxPAPC protects from septic shock by targeting the non-canonical inflammasome in macrophages [4].
Key features:
- A potent inhibitor of TLR2 and TLR4
- InvitroFit™: each lot is functionally tested
References:
1. Erridge C. et al., 2008. Oxidized Phospholipid Inhibition of Toll-like Receptor (TLR) Signaling Is Restricted to TLR2 and TLR4: roles for CD14, LPS-binding protein, and MD2 as targets for specificity of inhibition. J. Biol. Chem., 283: 24748-24759.
2. Stamenkovic A. et al., 2019. Oxidized lipids: not just another brick in the wall. Can J Physiol Pharmacol. 97(6):473-85.
3. von Schlieffen E. et al., 2009. Multi-Hit Inhibition of Circulating and Cell-Associated Components of the Toll-Like Receptor 4 Pathway by Oxidized Phospholipids. Arterioscler Thromb Vasc Biol, 29: 356-362.
4. Chu L.H. et al., 2018. The oxidized phospholipid oxPAPC protects from septic shock by targeting the non-canonical inflammasome in macrophages. Nat Commun. 9(1):996.
All products are for research use only, and not for human or veterinary use.
InvitroFit™
InvitroFit™ is a high-quality standard specifically adapted for in vitro studies of inhibitors. InvitroFit™ products are highly pure (≥95%) and guaranteed free of bacterial contamination, as confirmed using HEK Blue™ TLR2 and HEK Blue™ TLR4 cellular assays. Each lot is rigorously tested for functional activity using validated (or proprietary) cellular models. This grade ensures reliability and reproducibility for your research applications.
SPECIFICATIONS
Specifications
1 mg/ml in culture medium or chloroform
30 µg/ml for cell culture assays
Negative (tested using EndotoxDetect™ assay)
In vitro cellular assays
Each lot is functionally tested and validated using cellular assays.
CONTENTS
Contents
-
Product:OxPAPC
-
Cat code:tlrl-oxp1
-
Quantity:1 mg
OxPAPC is provided as a transparent film.
Shipping & Storage
- Shipping method: Room temperature
- -20°C
- Avoid repeated freeze-thaw cycles
Storage:
Caution:
Details
TLR2
Toll-like receptor 2 (TLR2) plays an essential role in detecting a diverse range of microbial pathogen-associated molecular patterns (PAMPs) from Gram-positive and Gram-negative bacteria as well as fungi, parasites, and viruses. These PAMPs include cell-wall components such as lipoproteins, lipoteichoic acid (LTA; Gram-positive bacteria only), lipoarabinomannan (mycobacteria only), and zymosan (yeast) [1]. Ligand recognition is enhanced by its non-specific delivery to TLR2 by CD14 [2, 3]. Upon ligand recognition, TLR2-dependent signaling cascades ultimately lead to a MyD88 and MAL/TIRAP-dependent activation of pro-inflammatory transcription factors such as NF-κB and AP-1 [4].
TLR4
Toll-like receptor 4 (TLR4) primarily recognizes and is activated by a core component of the outer membrane of Gram-negative bacteria, lipopolysaccharide (LPS). TLR4 requires interaction with a number of co-receptors including LPS-binding protein (LBP), CD14, and, myeloid differentiation protein 2 (MD-2) to bind to LPS and induce a signaling cascade [5, 6]. Ultimately, this leads to the activation of NF-κB and the production of pro-inflammatory cytokines.
References:
1. Oliveira-Nascimento L. et al., 2012. The Role of TLR2 in Infection and Immunity. Front Immunol 3, 79.
2. Jimenez-Dalmaroni M.J. et al., 2009. Soluble CD36 ectodomain binds negatively charged diacylglycerol ligands and acts as a co-receptor for TLR2. PLoS One 4, e7411.
3. Lotz S. et al., 2004. Highly purified lipoteichoic acid activates neutrophil granulocytes and delays their spontaneous apoptosis via CD14 and TLR2. J Leukoc Biol 75, 467-477.
4. Piao W. et al., 2016. Differential adapter recruitment by TLR2 co-receptors. Pathog Dis 74.
5. Cochet F. et al., 2017. The Role of Carbohydrates in the Lipopolysaccharide (LPS)/Toll-Like Receptor 4 (TLR4) Signalling. Int J Mol Sci. 18.
6. Kuzmich N.N. et al., 2017. TLR4 Signaling Pathway Modulators as Potential Therapeutics in Inflammation and Sepsis. Vaccines (Basel) 5.
Chemical structure of OxPAPC:
DOCUMENTS
Documents
Technical Data Sheet
Safety Data Sheet
Validation Data Sheet
Certificate of analysis
Need a CoA ?





