HEK-Dual™ hTLR4 Cells
-
Cat.code:
hkd-htlr4
- Documents
ABOUT
NF-κB–SEAP and IRF-Lucia reporter HEK293 cells expressing human TLR4
HEK-Dual™ hTLR4 cells were engineered from HEK-Dual™ cells, a human embryonic kidney HEK293-derived cell line, to study the Toll-like receptor 4 (TLR4)-dependent NF-κB and IRF responses upon stimulation with lipopolysaccharide (LPS). LPS is a major constituent of the outer membrane of Gram-negative bacteria and the main microbial mediator implicated in the pathogenesis of sepsis and septic shock [1].
Description
HEK-Dual™ hTLR4 cells feature the stable expression of TLR4, as well as the adapter proteins MD-2 (myeloid differentiation factor 2) and CD14 (cluster of differentiation 14). These cells also express two inducible reporter genes for SEAP (secreted embryonic alkaline phosphatase) and Lucia luciferase. As a result, these cells allow the simultaneous study of the NF-κB pathway, by monitoring the activity of SEAP, and the IRF pathway, by assessing the activity of the secreted Lucia luciferase. Upon TLR4 activation, both reporter proteins are readily measurable in the cell culture supernatant when using QUANTI-Blue™, a SEAP detection reagent, and QUANTI-Luc™ 4 Lucia/Gaussia, a Lucia luciferase detection reagent.
HEK-Dual™ hTLR4 cells are highly responsive to LPS. They show potent NF-κB and IRF responses upon incubation with TLR4-specific ligands, such as smooth LPS-EB Ultrapure (UP) or rough LPS-EK UP, when compared to HEK-Blue™ hTLR4 cells and the control cell line HEK-Dual™ (see figures).
Of note, HEK293 cells express endogenous levels of various PRRs, including TLR3 and TLR5, and therefore might respond to their cognate ligands (see figures).
Key features
- Stable expression of human TLR4, MD-2, and CD14
- Increased and reproducible response to rough and smooth LPS
- Distinct monitoring of TLR4-dependent NF-κB or IRF activation by assessing the SEA and& Lucia luciferase activities
Applications
- Detecting sample contamination with LPS
- Defining the role of TLR4 in LPS-induced signaling pathways
- Screening for TLR4 or antagonists
1. Godowski, P., 2005. A smooth operator for LPS responses. Nat Immunol 6, 544–546.
Disclaimer: These cells are for internal research use only and are covered by a Limited Use License (See Terms and Conditions). Additional rights may be available.
SPECIFICATIONS
Specifications
TLR4
Human
TLR4 activation cellular assays
Complete DMEM (see TDS)
Verified using Plasmotest™
Each lot is functionally tested and validated.
CONTENTS
Contents
-
Product:HEK-Dual™ hTLR4 Cells
-
Cat code:hkd-htlr4
-
Quantity:3-7 x 10^6 cells
- 1 ml Blasticidin (10 mg/ml)
- 1 ml Hygromycin B Gold (100 mg/ml)
- 1 ml Normocin™ (50 mg/ml)
- 1 ml Puromycin (10 mg/ml)
- 1 ml Zeocin® (100 mg/ml)
- 1 ml of QB reagent and 1 ml of QB buffer (sufficient to prepare 100 ml of QUANTI-Blue™ Solution, a SEAP detection reagent)
- 1 tube of QUANTI-Luc™ 4 Reagent, a Lucia luciferase detection reagent (sufficient to prepare 25 ml)
Shipping & Storage
- Shipping method: Dry ice
- Liquid nitrogen vapor
Storage:
Details
Toll-like receptor 4 signaling
The Toll-like receptor 4 (TLR4) was the first TLR identified and is an important pattern recognition receptor (PRR) in innate immunity and inflammation. It is found both on the cell surface and in endosomes of innate immune cells including monocytes and macrophages, as well as on intestinal epithelium and endothelial cells [1]. TLR4 can recognize pathogen- and damage-associated molecular patterns (PAMPs and DAMPs). However, it is primarily activated by lipopolysaccharide (LPS) and its toxic moiety Lipid A [2]. TLR4 does not directly interact with LPS, but requires essential adaptor proteins [3]. The soluble LPS-binding protein (LBP) extracts monomeric LPS from the microbial membrane and transfers it to CD14 (cluster of differentiation 14). This membrane-bound protein then interacts with MD-2 (myeloid differentiation factor 2), which is constitutively associated with the TLR4 ectodomain. The ligand-loaded MD-2 subsequently binds to another TLR4/MD-2/LPS complex, leading to their dimerization [4]. Then, TLR4 triggers two distinct signaling cascades [5]:
- the MyD88-dependent activating NF-κB pathway (at the cell surface)
- the TRIF-dependent activating IRF pathway (in endosomes)
At the cell surface, activation of TLR4 initiates the TIRAP-MyD88-dependent pathway, ultimately leading to the activation of NF-κB and the production of a pro-inflammatory response. Also, the TLR4 complex can be endocytosed into endosomes in a CD14-mediated fashion. This results in the the stimulation of IRF3 (interferon regulatory factor), which modulates the expression of type I IFN [3].
TLR4 signaling is crucial in both acute and chronic inflammatory disorders and thus, is an attractive target for novel treatments [1]. Stimulating drugs are useful for the development of vaccine adjuvants or cancer immunotherapeutics, whereas TLR4-inhibition is a therapeutic approach to treat septic shock or autoimmune inflammatory pathologies such as atherosclerosis [5].
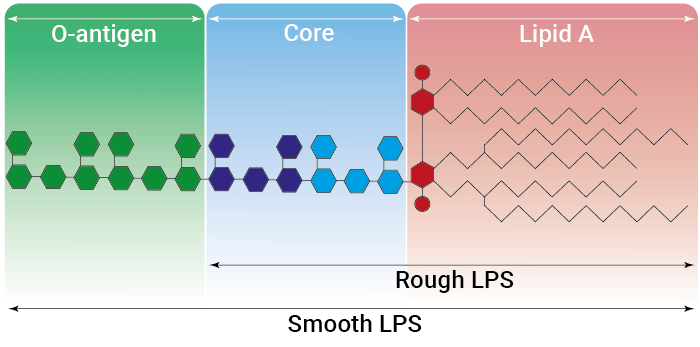
Rough vs smooth LPS
Lipopolysaccharide (LPS) is a major constituent of the outer membrane of Gram-negative bacteria. It comprises three covalently linked regions:
- the lipid A (endotoxin)
- the rough core oligosaccharide
- the O-antigenic side chain.
Wild-type LPS contains the O-side chain and is referred to as smooth (sLPS). Few bacterial strains (e.g. Salmonella Thyphimurium, Brucella canis) lost the O-side chain. This mutated form is called rough (rLPS). Both sLPS and rLPS share the same receptor complex (TLR4-MD-2-CD14), but their mechanism of action differs. While CD14 is necessary for sLPS NF-κB and IRF signaling, it is dispensible for rLPS NF-κB signaling. It has been hypothesized that rLPS activates a broader range of cells (CD14 positive, low and negative), accounting for higher toxicity [6].
Nevertheless, both LPS variations elicit potent innate immune responses. The resulting signaling triggers the release of pro‑inflammatory cytokines, which can lead to both acute and chronic inflammatory diseases. It is all about balance: small controlled amounts of LPS can be protective and large uncontrolled amounts can lead to disastrous outcomes, such as septic shock [7]. Despite its highly inflammatory nature, LPS has remarkable therapeutic potential and features many characteristics needed for an effective vaccine adjuvant [8].
References:
1. Ou, T. et al. 2018. The Pathologic Role of Toll-Like Receptor 4 in Prostate Cancer. Front Immunol 9, 1188.
2. Cochet, F. et al. 2017. The Role of Carbohydrates in the Lipopolysaccharide (LPS)/Toll-Like Receptor 4 (TLR4) Signalling. Int J Mol Sci 18
3. Kuzmich, N.N. et al. 2017. TLR4 Signaling Pathway Modulators as Potential Therapeutics in Inflammation and Sepsis. Vaccines (Basel) 5.
4.Tanimura N. et al. 2014. The attenuated inflammation of MPL is due to the lack of CD14-dependent tight dimerization of the TLR4/MD2 complex at the plasma membrane. Int Immunol.(6):307-14.
5. Romerio A, Peri F. 2020. Increasing the Chemical Variety of Small-Molecule-Based TLR4 Modulators: An Overview. Front Immunol.;11:1210.
6. Zanoni I,.et al., 2012. Similarities and differences of innate immune responses elicited by smooth and rough LPS. Immunol Lett. 2012 Feb 29;142(1-2):41-7.
7. Godowski, P., 2005. A smooth operator for LPS responses. Nat Immunol 6, 544–546.
8. McAleer, J.P. & Vella, A.T., 2010. Educating CD4 T cells with vaccine adjuvants: lessons from lipopolysaccharide. Trends Immunol 31, 429-435.
DOCUMENTS
Documents
Technical Data Sheet
Validation Data Sheet
Safety Data Sheet
Certificate of analysis
Need a CoA ?









