2’3’-c-di-AM(PS)2 (Rp,Rp) VacciGrade™
-
Analog of c-di-AMP
-
Cat.code:
vac-nacda2r
- Documents
ABOUT
2’3’-c-diAM(PS)2(Rp,Rp) endotoxin-free, sterile STING ligand
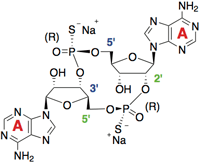
2’3’-c-di-AM(PS)2 (Rp,Rp) is the Rp, Rp-isomer of the 2’3’ bisphosphorothioate analog of 3’3’-cyclic adenosine monophosphate (c-di-AMP), a cyclic dinucleotide (CDN). c-di-AMP is a second messenger molecule produced by bacteria and has potent immunostimulant activity in mammals [1]. It activates innate immunity by directly binding the endoplasmic reticulum-resident receptor STING (stimulator of interferon genes), leading to the expression of interferon-β (IFN-β) and nuclear factor-κB (NF-κB) dependent inflammatory cytokines [2].
Key Features
- 2’3’-c-di-AM(PS)2 (Rp,Rp) has a higher affinity for STING than c-di-AMP due to the presence of a 2’-5’, 3’-5’ mixed linkage, as found in endogenous human CDNs produced by cGAS (cyclic GMP-AMP (cGAMP) synthase) [3]. It activates all known human STING alleles, as well as mouse STING.
- In addition, this c-di-AMP analog contains two phosphorothioate diester linkages, which prevent its degradation by the phosphodiesterases that are present in host cells or in the systemic circulation [4].
- Finally, this Rp, Rp dithio diastereoisomer was found to induce higher type I IFN production when compared to the Rp/Sp dithio diastereoisomers or c-di-AMP [3].
2’3’-c-di-AM(PS)2 (Rp,Rp) VacciGrade™ is a high-quality pre-clinical grade.
![]() Read our review on STING: Deciphering the STING Paradox
Read our review on STING: Deciphering the STING Paradox
References:
1. Woodward JJ. et al., 2010. c-di-AMP secreted by intracellular Listeria monocytogenes activates a host type I interferon response. Science.328(5986):1703-5.
2. Jin L. et al., 2011. MPYS is required for IFN response factor 3 activation and type I IFN production in the response of cultured phagocytes to bacterial second messengers cyclic-di-AMP and cyclic-di- GMP. J Immunol. 187(5):2595-601.
3. Corrales L et al., 2015. Direct activation of STING in the tumor microenvironment leads to potent and systemic tumor regression and immunity. Cell Rep. 11(7):1018-30.
4. Yan H. et al., 2008. Synthesis and immunostimulatory properties of the phosphorothioate analogs of cdiGMP. Bioorg. Med. Chem. Lett. 18, 5631–5634.
All products are for internal research use only, and not for human or veterinary use.
VacciGrade™
VacciGrade™ is a high-quality pre-clinical grade, suitable for in vivo studies. VacciGrade™ products are filter-sterilized (0.2 µm) and filled under strict aseptic conditions in a clean room. The absence of bacterial contamination is assessed by a sterility test using a pharmacopeia-derived assay.
SPECIFICATIONS
Specifications
STING agonist
STING
C20H22N10O10P2S2 •2Na
50 mg/ml in water
0.2 µm filtration, Sterility guaranteed
< 5 EU/mg (determined using the HEK-Blue™ LPS Detection Kit 2)
Induction of interferon pathway in cellular assays
Each lot is functionally tested and validated using cellular assays
CONTENTS
Contents
-
Product:2’3’-c-di-AM(PS)2 (Rp,Rp) VacciGrade™
-
Cat code:vac-nacda2r
-
Quantity:500 µg
10 ml sterile endotoxin-free physiological water (NaCl 0.9%)
Shipping & Storage
- Shipping method: Room temperature
- -20°C
- Avoid repeated freeze-thaw cycles
Storage:
Caution:
Details
Structure of 23-cdiAMPS2
DOCUMENTS
Documents
Technical Data Sheet
Validation Data Sheet
Safety Data Sheet
Certificate of analysis
Need a CoA ?





