Anti-hIFN-γ Neutralizing mAb
| Product | Unit size | Cat. code | Docs. | Qty. | Price | |
|---|---|---|---|---|---|---|
|
Anti-hIFN-γ-IgA Neutralizing recombinant IgA2 mAb against human IFN-γ (clone H7WM120) |
Show product |
200 µg |
hifng-mab7-02
|
|
Neutralizing IgA monoclonal antibody to human interferon-gamma

Neutralizing monoclonal antibody against human IFN-γ
Anti-hIFN-γ-hIgA2 (clone H7WM120) is a recombinant monoclonal antibody (mAb) able to neutralize human interferon-gamma (hIFN-γ). This mAb is expressed and produced in chinese hamster ovary (CHO) cells, ensuring reliability and lot-to-lot reproducibility. Thereby, common hybridoma-related drawbacks such as the generation of non-relevant mAbs containing aberrant light chains are avoided [1].
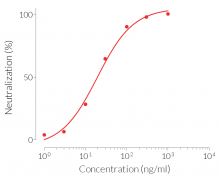
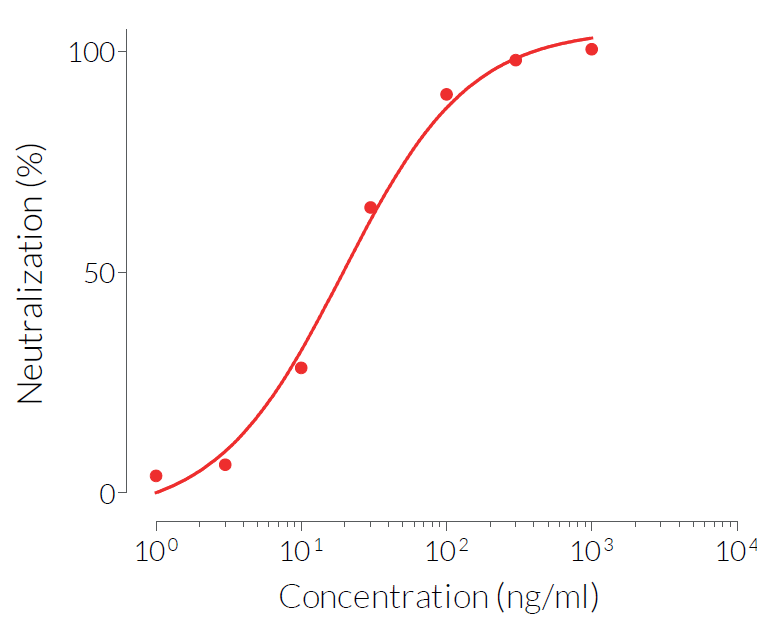
Anti-hIFN-γ-hIgA2 consists of the variable region of the Anti-hIFN-γ mAb (clone H7WM120) and the engineered human IgA2 constant region. This antibody has been selected for its ability to efficiently neutralize the biological activity of hIFN-γ. It was found to have significantly higher neutralizing activity than its corresponding IgG isotype.
Key features:
- Potent and specific neutralization activity against hIFN-γ
- Free from non-relevant mAbs found in hybridoma-based productions
- Provided azide-free
- Each lot is functionally tested
IFN-γ plays a role in activating lymphocytes to enhance anti-microbial and anti-tumor effects [2-4].
References:
1. Bradbury A. et al. 2018. When monoclonal antibodies are not monospecific: Hybridomas frequently express additional functional variable regions. mAbs, 10(4), 539–546.
2. Ivashkiv L.B., 2018. IFNγ: signalling, epigenetics and roles in immunity, metabolism, disease and cancer immunotherapy. Nat Rev Immunol. 18(9):545-558.
3. Shtrichman R. & Samuel CE., 2001. The role of gamma interferon in antimicrobial immunity. Curr Opin Microbiol. 4(3):251-9.
4. Sato A. et al., 2006. Antitumor activity of IFN-lambda in murine tumor models. J Immunol. 176(12):7686-94.
Specifications
Target: Human interferon-gamma (hIFN-γ)
Source: CHO cells
Clone: H7WM120
Isotype: Human IgA2, kappa
Control: Human IgA2 Control
Purification: Affinity chromatography with peptide M
Formulation: 0.2 µm filtered solution in Tris HCl buffer with saccharose, glycine, and stabilizing agents
Applications: Blocking & Neutralization
Quality control:
- This product have been validated using neutralization cellular assays.
- The complete sequence of the antibody has been verified.
- The absence of bacterial contamination (e.g. lipoproteins and endotoxins) has been confirmed using HEK-Blue™ TLR2 and HEK‑Blue™ TLR4 cells.
Contents
- 200 μg purified anti-hIFNγ-IgA antibody, provided azide-free and lyophilized.
![]() The product is shipped at room temperature.
The product is shipped at room temperature.
![]() Store lyophilized antibody at -20 °C.
Store lyophilized antibody at -20 °C.
![]() Lyophilized product is stable for at least 1 year.
Lyophilized product is stable for at least 1 year.
![]() Avoid repeated freeze-thaw cycles.
Avoid repeated freeze-thaw cycles.
Details
Interferon-gamma (IFN-γ), a Type II interferon, is secreted from CD4+ T-helper 1 (Th1) cells and activated natural killer (NK) cells. It plays a role in activating lymphocytes to enhance anti-microbial and anti-tumor effects [1-3]. In addition, IFN-γ plays a role in regulating the proliferation, differentiation, and response of lymphocyte subsets.
IFN-γ exerts its action by first binding to a heterodimeric receptor consisting of two chains, IFNGR1 and IFNGR2, causing its dimerization and the activation of specific Janus family kinases (JAK1 and JAK2) [4, 5]. Two STAT1 molecules then associate with this ligand-activated receptor complex and are activated by phosphorylation. Activated STAT1 forms homodimers and are translocated to the nucleus where they bind interferon-gamma-activated sites (GAS) in the promoter of IFN-γ inducible genes.
1. Ivashkiv L.B., 2018. IFNγ: signalling, epigenetics and roles in immunity, metabolism, disease and cancer immunotherapy. Nat Rev Immunol. 18(9):545-558.
2. Shtrichman R. & Samuel CE., 2001. The role of gamma interferon in antimicrobial immunity. Curr Opin Microbiol. 4(3):251-9.
3. Sato A. et al., 2006. Antitumor activity of IFN-lambda in murine tumor models. J Immunol. 176(12):7686-94.
4. Platanias L.C., 2005. Mechanisms of type-I- and type-II-interferon-mediated signalling. Nat Rev Immunol. 5(5):375-86.
5. Schroder K. et al., 2004. Interferon-gamma: an overview of signals, mechanisms, and functions. J Leukoc Biol. 75(2):163-89.