THP1-Dual KO-STING Cells
-
Cat.code:
thpd-kostg
- Documents
ABOUT
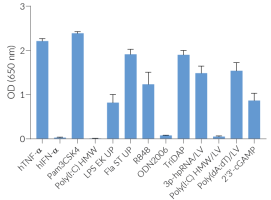
STING knockout dual reporter monocytes
THP1-Dual™ KO-STING cells were generated from THP1-Dual™ cells by stable knockout of the STING gene. STING (stimulator of interferon genes), alternatively known as MPYS, TMEM173, MITA, and ERIS is a direct sensor of cyclic dinucleotides (CDNs) [1-3]. These cell lines were derived from human THP‑1 monocytes, a cell line often used to study DNA sensing pathways as they express all the cytosolic DNA sensors identified so far (with the exception of DAI).
THP1-Dual™ and THP1-Dual™ KO-STING cells stably express two inducible secreted reporter genes: IFN-inducible Lucia luciferase and NF‑κB-inducible SEAP (secreted embryonic alkaline phosphatase). They can be used to study the role of STING by monitoring IRF-induced Lucia luciferase activity, using QUANTI‑Luc™ 4 Lucia/Gaussia, a Lucia and Gaussia luciferase detection reagent.
THP1-Dual™ KO-STING cells are resistant to the selectable markers blasticidin and Zeocin®.
References:
1. Burdette DL. et al., 2011. STING is a direct innate immune sensor of cyclic di-GMP. Nature. 478(7370):515-8.
2. Zhang X. et al., 2013. Cyclic GMP-AMP containing mixed phosphodiester linkages is an endogenous high-affinity ligand for STING. Mol Cell. 51(2):226-35.
3. Barber GN. et al., 2015. STING: infection, inflammation and cancer. Nat Rev Immunol. 15(12):760-70.
Disclaimer: These cells are for internal research use only and are covered by a Limited Use License (See Terms and Conditions). Additional rights may be available.
SPECIFICATIONS
Specifications
Negative control for STING activation cellular assays
Complete RPMI 1640 (see TDS)
Verified using Plasmotest™
Each lot is functionally tested and validated.
CONTENTS
Contents
-
Product:THP1-Dual KO-STING Cells
-
Cat code:thpd-kostg
-
Quantity:3-7 x 10^6 cells
- 1 ml of Normocin™ (50 mg/ml). Normocin™ is a formulation of three antibiotics active against mycoplasmas, bacteria and fungi
- 1 ml of Zeocin® (100 mg/ml)
- 1 ml of Blasticidin (10 mg/ml)
- 1 tube of QUANTI-Luc™ 4 Reagent, a Lucia luciferase detection reagent (sufficient to prepare 25 ml)
- 1 ml of QB reagent and 1 ml of QB buffer (sufficient to prepare 100 ml of QUANTI-Blue™ Solution, a SEAP detection reagent)
Shipping & Storage
- Shipping method: Dry ice
- Liquid nitrogen vapor
Storage:
DOCUMENTS
Documents
Technical Data Sheet
Validation Data Sheet
Safety Data Sheet
Certificate of analysis
Need a CoA ?