THP1-Dual™ KO-IFNAR2 Cells
-
Cat.code:
thpd-koifnar2
- Documents
ABOUT
IFNAR2 knockout NF-κB-SEAP and IRF-Lucia luciferase reporter monocytes
THP1-Dual™ KO-IFNAR2 cells were generated from THP1-Dual™ cells by stable knockout of the IFNAR2 gene. They derive from human THP‑1 monocytes, a cell line often used to study DNA sensing pathways as they express all the cytosolic DNA sensors identified so far (with the exception of DAI).
THP1-Dual™ and THP1-Dual™ KO-IFNAR2 cells stably express two inducible secreted reporter genes: Lucia luciferase and SEAP (secreted embryonic alkaline phosphatase). The Lucia luciferase reporter gene is under the control of an ISG54 (interferon-stimulated gene) minimal promoter in conjunction with five IFN-stimulated response elements. The SEAP gene is driven by an IFN-β minimal promoter fused to five copies of the NF‑kB response element. As a result, they allow the simultaneous study of the IFN regulatory factor (IRF) pathway, by assessing the activity of Lucia luciferase and the NF-kB pathway, by monitoring the activity of SEAP. Both reporter proteins are readily measurable in the cell culture supernatant when using QUANTI‑Luc™ 4 Lucia/Gaussia, a Lucia luciferase detection reagent, and QUANTI-Blue™, a SEAP detection reagent.
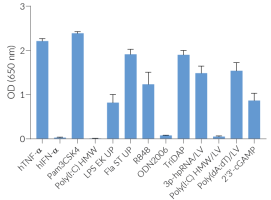
THP1-Dual™ KO-IFNAR2 and THP1-Dual™ cells can be used to study the role of IFNAR2 by monitoring IRF-induced Lucia luciferase activity. THP1-Dual™ KO-IFNAR2 cells are resistant to blasticidin and Zeocin®.
References:
1. Zao W. et al., 2008. A conserved IFN-alpha receptor tyrosine motif directs the biological response to type I IFNs. J Immunol. 180(8):5483-9.
2. de Weerd NA. et al., 2007. Type I interferon receptors: biochemistry and biological functions. J Biol Chem. 282(28):20053-7.
Disclaimer: These cells are for internal research use only and are covered by a Limited Use License (See Terms and Conditions). Additional rights may be available.
SPECIFICATIONS
Specifications
Dual reporter assays for IFNAR2 knockout, NF-κB and IRF signaling pathways
Complete RPMI 1640 (see TDS)
Verified using Plasmotest™
Each lot is functionally tested and validated.
CONTENTS
Contents
-
Product:THP1-Dual™ KO-IFNAR2 Cells
-
Cat code:thpd-koifnar2
-
Quantity:3-7 x 10^6 cells
- 1 ml of Normocin™ (50 mg/ml)
- 1 ml of Zeocin® (100 mg/ml)
- 1 ml of Blasticidin (10 mg/ml)
- 1 tube of QUANTI-Luc™ 4 Reagent
- 1 ml of QB reagent and 1 ml of QB buffer
See Technical Data sheet for storage information
Shipping & Storage
- Shipping method: Dry ice
- Liquid nitrogen vapor
- Upon receipt, store immediately in liquid nitrogen vapor. Do not store cell vials at -80°C.
Storage:
Caution:
Details
THP1 reporter cells are a family of cells derived from the human monocytic THP-1 cell line, which naturally expresses many pathogen recognition receptors (PRRs), including Toll-like receptors.
They respond to ligands for certain TLRs; namely, TLR2, TLR1/2, TLR2/6, TLR4, TLR5 and TLR8. These cells can also be used to study DNA sensing pathways, as they are highly responsive to PRR agonists that trigger interferon (IFN) signaling pathways.
THP1‑Dual™ cells feature two reporter genes that enable the simultaneous study of the NF-κB and IFN signaling pathways.
Type I interferons (IFNs), in particular IFN-α and IFN-β, are vital for host resistance to viral infections. Their activity is mediated by the IFN-α receptor (IFNAR), which consists of two chains: IFNAR1 and IFNAR2. Following their production, these cytokines bind IFNAR and recruit the Janus kinases (JAK1 and TyK2), leading to the phosphorylation of STAT1 and STAT2, which then dimerize. These dimers interact with IFN regulatory factor 9 (IRF9), forming a complex named ISGF3, which binds to IFN-stimulated response elements (ISRE) in the promoters of IFN-stimulated genes (ISG) to regulate their expression. Knockout of the IFNAR2 gene abolishes the cellular response to type I IFNs [1,2].
DOCUMENTS
Documents
Technical Data Sheet
Validation Data Sheet
Safety Data Sheet
Certificate of analysis
Need a CoA ?