cAIMP Difluor
-
Cat.code:
tlrl-nacaidf-05
- Documents
ABOUT
cAIMP difluorinated
InvivoGen's cAIMP Difluor (also known as CL614) is a derivative of cAIMP, an analog of the bacterial cyclic dinucleotide (CDN) 3’3’-cGAMP [1].
cAIMP and cAIMP Difluor are novel STING (stimulator of interferon genes)-activating synthetic CDNs.
Unlike natural CDNs, whose constituent nucleosides are guanosine and/or adenine, cAIMP and its derivatives contain one adenine nucleoside and one inosine nucleoside.
cAIMP Difluor is composed of two 2’-deoxynucleosides with a fluorine atom at 2’ position of each nucleoside.
STING ligands such as cAIMP induce the production of type I interferons (IFNs) and of proinflammatory cytokines through the IRF and NF-κB pathways, respectively.
The incorporation of fluorine into biologically active molecules is commonly used in medicinal chemistry to improve their metabolic stability or to modulate physicochemical properties such as lipophilicity [2]. Moreover, the introduction of a fluorine atom can change the biological activities of a molecule. Indeed cAIMP
Difluor (referred to as compound 52 by Lioux et al. [1]) more potently induces interferon regulatory factor (IRF) and NF-κB pathways in a STING‑dependent manner when compared to STING agonists such as 2’3’‑cGAMP and DMXAA [1]. Interestingly, cAIMP Difluor is more resistant than 2’3’‑cGAMP and cAIMP to cleavage by certain nuclease and phosphodiesterase enzymes [1].
To facilitate the srudy of STING ligands, InvivoGen has developed stable reporter cells in two well established immune cell models: THP-1 human monocytes and RAW 264.7 murine macrophages. These cells express inducible SEAP and/or Lucia luciferase reporter genes under the control of an IRF-inducible and NF-κB promoter.
References:
1. Lioux T. et al., 2016. Design, synthesis, and biological evaluation of novel cyclic adenosine-inosine monophosphate (cAIMP) analogs that activate stimulator of interferon genes (STING). J Med Chem. 59(22):10253-10267.
2. Böhm HJ. et al., 2004. Fluorine in medicinal chemistry. Chembiochem. 5(5):637-43
All products are for research use only, and not for human or veterinary use.
SPECIFICATIONS
Specifications
STING
C20H21F2N9O11P2 • 2Na
100 ng - 30 µg/ml
50 mg/ml in water
0.2 µm filtration
Negative (tested using EndotoxDetect™ assay)
Cellular assays
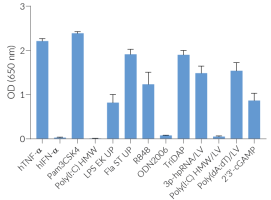
Each lot is functionally tested and validated using THP1-Dual™ Cells.
CONTENTS
Contents
-
Product:cAIMP Difluor
-
Cat code:tlrl-nacaidf-05
-
Quantity:2 x 250 µg
1.5 ml endotoxin-free water
Shipping & Storage
- Shipping method: Room temperature
- Upon receipt, store product at -20 °C
- Avoid repeated freeze-thaw cycles
Storage:
Caution:
DOCUMENTS
Documents
Technical Data Sheet
Safety Data Sheet
Validation Data Sheet
Certificate of analysis
Need a CoA ?