FSL-1
-
Cat.code:
tlrl-fsl
- Documents
ABOUT
Synthetic diacylated lipoprotein
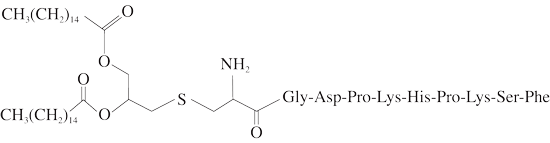
FSL-1 (Pam2CGDPKHPKSF) is a synthetic lipoprotein derived from Mycoplasma salivarium [1]. It contains the same diacylglycerol structure as MALP-2 from M. fermentans but has a different peptide sequence [2]. The mycoplasmal lipoprotein FSL-1 contains a diacylated cysteine residue and is recognized by the TLR2/TLR6 heterodimer [3].
Mode of action:
At the cell surface, TLR2 forms a heterodimer with co-receptors TLR1 or TLR6, depending upon either tri- or diacylation of the ligand. Once a ligand binds to either TLR2-TLR1 or TLR2-TLR6, a MyD88-dependent activation of NF-κB and AP-1 occurs, ultimately leading to an innate immune response.
Recognition of FSL-1, a diacylated lipoprotein, is mediated by TLR2 which cooperates with TLR6 through their cytoplasmic domain to induce a signaling cascade leading to AP-1 and NF-κB activation and cytokine production [3,4].
Key features of FSL-1:
- Potent activator of the TLR2/TLR6 heterodimer
- Synthetic lipoprotein free of microbial contaminants
- Each lot is functionally tested
References:
1. Shibata K.I. et al., 2000. The N-terminal lipopeptide of a 44-kDa membrane-bound lipoprotein of Mycoplasma salivarium is responsible for the expression of intercellular adhesion molecule-1 on the cell surface of normal human gingival fibroblasts. J. Immunol. 165:6538–44.
2. Okusawa T. et al., 2004. Relationship between structures and biological activities of mycoplasmal diacylated lipopeptides and their recognition by Toll-Like receptors 2 and 6. Infect Immun. 72(3): 1657-1665.
3. Takeuchi O. et al., 2001. Discrimination of bacterial lipoproteins by Toll-like receptor 6. Int Immunol. 13(7):933-40.
4. Ahmad R. et al., 2014. FSL-1 induces MMP-9 production through TLR-2 and NF-κB /AP-1 signaling pathways in monocytic THP-1 cells. Cell Physiol Biochem. 34(3):929-42.
All products are for research use only, and not for human or veterinary use.
SPECIFICATIONS
Specifications
C84H140N14O18S
10 pg/ml - 100 ng/ml
10 mg/ml in water
Activation of TLR2 confirmed using cellular assays
Activation of TLR2 confirmed using cellular assays
CONTENTS
Contents
-
Product:FSL-1
-
Cat code:tlrl-fsl
-
Quantity:100 µg
1.5 ml sterile endotoxin-free water
Shipping & Storage
- Shipping method: Room temperature
- 4°C
- Avoid repeated freeze-thaw cycles
Storage:
Caution:
Details
TLR2
TLR2 plays a pivotal role in detecting a diverse range of pathogen-associated molecular patterns (PAMPs) from bacteria, viruses, fungi, and parasites [1].
Specifically, it recognizes cell-wall components including lipoproteins and peptidoglycan from bacteria, lipoteichoic acid from Gram-positive bacteria, lipoarabinomannan from mycobacteria, and zymosan from fungi. Notably, at the cell surface TLR2 forms a heterodimer with co-receptors TLR1 or TLR6, depending upon either tri- or diacylation of the ligand, respectively.
Downstream signaling of both TLR2 heterodimers has been shown to be enhanced when in association with CD14 [2]. Once a ligand binds to either TLR2-TLR1 or TLR2-TLR6, a MyD88- dependent activation of NF-κB and AP-1 occurs, ultimately leading to an innate immune response. Importantly, the ability for TLR2 to form heterodimers not only expands the range of PAMPs that it recognizes but can also lead to divergent responses depending on the heterodimer involved [3]. To date, TLR2 alone as a functional homodimer has only been proposed, with no evidence to prove that it triggers a signaling cascade [1].
Reference:
1. Oliveira-Nascimento L. et al., 2012. The Role of TLR2 in Infection and Immunity. Front Immunol. 3:79.
2. Lotz S. et al., 2004. Highly purified lipoteichoic acid activates neutrophil granulocytes and delays their spontaneous apoptosis via CD14 and TLR2. J Leukoc Biol. 75(3):467-77.
3. Nguyen M.T. et al., 2017. Lipid moieties on lipoproteins of commensal and non-commensal staphylococci induce differential immune responses. Nat Commun. 8(1):2246.
Chemical structure of FSL-1:
DOCUMENTS
Documents
Technical Data Sheet
Validation Data Sheet
Safety Data Sheet
Certificate of analysis
Need a CoA ?




