MycoStrip® - Mycoplasma Detection Kit
| Product | Unit size | Cat. code | Docs. | Qty. | Price | |
|---|---|---|---|---|---|---|
|
MycoStrip® Mycoplasma Detection Kit |
Show product |
10 tests 20 tests 50 tests |
rep-mys-10
|
|||
|
MycoStrip® 50 Mycoplasma Detection Kit (WITHOUT CASSETTE) |
Show product |
50 tests |
rep-mysnc-50
|
|||
|
MycoStrip® 100 Mycoplasma Detection Kit (WITHOUT CASSETTE) |
Show product |
100 tests |
rep-mysnc-100
|
Rapid mycoplasma detection with MycoStrip®
Easy and fast detection of mycoplasma contamination in cell cultures
With over 40 years of experience in developing mycoplasma solutions for the scientific community, InvivoGen's MycoStrip® offers a new method for the detection of mycoplasma in cell culture, allowing you to combat the contamination.
MycoStrip® is a simple test that requires no special lab equipment, is highly sensitive and provides clear results, with no false positives. Furthermore, with results in less than an hour, MycoStrip® allows you to swiftly start treating the contaminated cell culture.
What if my test is positive?
Don't stress! Your culture is easily treatable with InvivoGen's anti-mycoplasma reagents.
Treat your culture and eradicate the contamination using Plasmocin® or Plasmocure™. Upon completion of the treatment (~2 weeks), re-test using MycoStrip®.
Assay Principle
Detection of cell culture contaminating mycoplasma by MycoStrip® is based on isothermal PCR. Simply prepare your sample and add our proprietary Reaction Mix to target and amplify the 16S rRNA gene for the most commonly found mycoplasma species in cell culture. Results are clearly visualized on an immunochromatographic strip within 5 minutes.
Key features of MycoStrip®
➔ Simple
Easy to perform, and unlike most other mycoplasma detection assays, no special lab equipment is required.
➔ Rapid
Performed in 1 hour, with less than 15 minutes of hands-on time.
➔ Clear
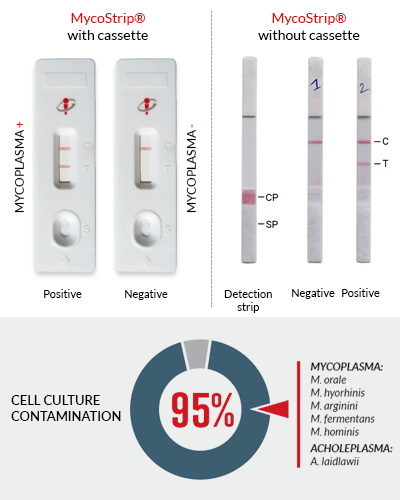
Fast and easy-to-interpret results that appear within 2-5 minutes, as 1 or 2 bands on the strip.
- One band - negative for mycoplasma
- Two bands - positive for mycoplasma
➔ Specific
MycoStrip® has been specifically designed to detect the Mycoplasma and Acholeplasma species that most commonly contaminate cell culture. These include the six species that account for 95% of all contaminations: M. orale, M. hyorhinis, M. arginini, M. fermentans, M. hominis, and A. laidlawii. Importantly, because the MycoStrip® reaction mix detects unique regions of DNA there is no cross‑reactivity with other bacterial, fungal or mammalian DNA.
➔ Sensitive
MycoStrip® is able to detect as low as 10-102 CFU/ml and thus, can be used to detect mycoplasma contamination before it significantly affects experimental results, which typically occurs at ~107 CFU/ml [1].
➔ Also available without cassettes
The larger unit sizes (50 and 100 strips) are available without cassettes, offering a more economical and eco-friendly version of Mycostrip®.
![]() Read the comparative study on mycoplasma detection methods from the Japanese Cell Bank
Read the comparative study on mycoplasma detection methods from the Japanese Cell Bank
Note: MycoStrip® is a trademark of InvivoGen registered with the United States Patent and Trademark Office and foreign equivalents.
All InvivoGen products are for internal research use only, and not for human or veterinary use.
References:
1. Drexler, H.G. & Uphoff, C.C. 2002. Mycoplasma contamination of cell cultures: Incidence, sources, effects, detection, elimination, prevention. Cytotechnology 39, 75-90.
Back to the topContents
MycoStrip® contains:
| With cassette | Without cassette | ||||
| Contents | 10 Tests | 20 Tests | 50 Tests | 50 Tests | 100 Tests |
|
• Reaction Mix • Reaction Buffer • Migration Buffer • Positive Control • Detection strips |
1 vial 1 vial 2 vials 1 vial 10 |
2 vials 2 vials 3 vials 1 vial 20 |
5 vials 4 vials 1 bottle 1 vial 50 |
5 vials 4 vials 1 bottle 1 vial 50 |
10 vials 8 vials 2 bottles 2 vials 100 |
![]() MycoStrip® is shipped at room temperature.
MycoStrip® is shipped at room temperature.
![]() Store all components (including the detection strips) at -20°C.
Store all components (including the detection strips) at -20°C.
![]() If properly stored, all components are stable for 12 months.
If properly stored, all components are stable for 12 months.
Specifications
Acholeplasma & Mycoplasma strains detected by MycoStrip®
MycoStrip® has been specifically designed to detect Mycoplasma and Acholeplasma species that most commonly contaminate cell cultures (see below). Importantly, there is no cross-reactivity with any other tested bacterial, fungal, or mammalian DNA when using MycoStrip®.
Mollicutes
|
Bacteria & Yeast
|
||
| Mycoplasma gallisepticum | + | Pseudomonas aeruginosa | - |
| Mycoplasma fermentans | + | Staphylococcus aureus | - |
| Mycoplasma arginini | + | Staphylococcus epidermidis | - |
| Mycoplasma orale | + | Bacillus cereus | - |
| Mycoplasma hyorhinis | + | Escherichia coli | - |
| Mycoplasma hominis | + | Listeria monocytogenes | - |
| Mycoplasma salivarium | + | Salmonella sp. | - |
| Mycoplasma conjunctivae | + | Candida albicans | - |
| Mycoplasma spumans | + | ||
| Mycoplasma arthritidis | + | ||
| Mycoplasma bovirhinis | + | ||
| Mycoplasma synoviae | + | ||
| Mycoplasma pulmonis | + | ||
| Mycoplasma alkalescens | + | ||
| Mycoplasma buccale | + | ||
| Mycoplasma faucium | + | ||
| Mycoplasma canadense | + | ||
| Mycoplasma hyopneumoniae | + | ||
| Mycoplasma hyosynoviae | + | ||
| Mycoplasma lipophilum | + | ||
| Mycoplasma bovis | + | ||
| Mycoplasma neurolyticum | + | ||
| Mycoplasma spermatophilum | + | ||
| Mycoplasma primatum | + | ||
| Mycoplasma pneumoniae | + | ||
| Mycoplasma leachii | + | ||
| Mycoplasma amphoriforme | + | ||
| Mycoplasma bovoculi | + | ||
| Mycoplasma californicum | + | ||
| Mycoplasma genitalium | + | ||
| Mycoplasma haematobovis | + | ||
| Mycoplasma haemohominis | + | ||
| Mycoplasma haemosuis | + | ||
| Mycoplasma mycoïdes | + | ||
| Mycoplasma pirum | + | ||
| Mycoplasma wenyonii | + | ||
| Mycoplasma muris | + | ||
| Acholeplasma laidlawii | + | ||
| Acholeplasma modicum | + | ||
| Acholeplasma morum | + | ||
| Ureaplasma parvum | + |
Details
MYCOPLASMA CONTAMINATION
Mycoplasma is a major problem in cell culture, with various sources of contamination such as lab operators, dirty water baths/incubators, and biological reagents. Due to their small size, mycoplasma is undetectable by visual inspection, they pass through standard filtration, and are resistant to a large number of antibiotics. There are many reasons to regularly test your cell culture for mycoplasma and these include:
- Loss of precious samples
- Serious impact on data reliability and reproducibility
- Testing is required by most journals for publication
- Loss of time and money
More information on mycoplasma in cell culture
Back to the topFAQs
1. What if my test is positive for mycoplasma?
InvivoGen has the solution! Your culture is easily treatable with our comprehensive anti-mycoplasma reagents. Simply treat your culture with Plasmocin® or Plasmocure™ and upon completion of the treatment (~2 weeks), re-test using MycoStrip®.
Note: For more information check out our Practical Guide to Cell culture contamination.
2. I have collected some cells with the supernatant that is to be tested, will this interfere with the assay?
There is no need to worry, the presence of mammalian cell DNA will not affect the detection assay.
3. During sample preparation, is there an advantage to heating the sample to 95°C?
There is no increase in sensitivity however this step will kill the mycoplasma avoiding the handling of live mycoplasma.
4. Is it necessary to include a positive and negative control every time when using MycoStrip®?
It is important to include positive and negative controls on a regular basis to monitor the reliability of your results and in case of troubleshooting.
5. At what stage can I freeze my sample for later analysis?
Samples can be stored as followed:
- Cell culture supernatant: before starting the “preparation of samples” protocol, you can store the supernatant at -20°C until required. Before use, completely thaw the sample at 37°C and mix well by vortexing to ensure all salts are dissolved.
- Prepared sample: After performing steps 1 – 4 of the “preparation of samples” protocol, your sample can be store at -20°C until required.
- Processed sample: After performing steps 1 – 4 of the “detection assay” protocol, the processed samples can be stored at -20°C for up to 3 months.
6. I have a faint test band for one of my samples and am unsure if this should be considered as positive for mycoplasma contamination. What would you recommend?
If you observe a faint band for one of your samples, we consider this to be a positive result for mycoplasma contamination. We recommend confirming this result using one of the following tips:
- Concentrate your sample: begin with a larger volume of supernatant and/or repeat steps 2-3 in the “preparation of samples” protocol. Re‑test the concentrated sample using MycoStrip®.
- Continue to grow the culture for an additional 48 hours before re-testing using MycoStrip®.
- Use sterile water (nuclease-free) to resuspend the sample instead of PBS (to increase the sensitivity of the test).
7. I do not have a thermocycler in the lab, what would you recommend to use instead?
If you do not have a thermocycler, you can use a calibrated heatblock or a water bath instead. Please note that the use of a water bath can introduce new sources of contamination.
8. I have left my prepared sample for over an hour on the heat block and wonder if I can still use this sample?
Exceeding the 40-minute incubation at 65°C significantly impacts the results of the assay and therefore, we do not recommend to use this sample.