Recombinant anti-mouse CD25 antibodies
| Product | Unit size | Cat. code | Docs. | Qty. | Price | |
|---|---|---|---|---|---|---|
|
Anti-mCD25-PC-mIgG2a InvivoFit™ PC-61.5.3-derived monoclonal antibody against murine CD25 (blocking) |
Show product |
1 mg 10 mg |
mcd25c1-mab10-1
|
|||
|
Anti-mCD25-7D4-mIgG2a InvivoFit™ 7D4-derived monoclonal antibody against murine CD25 (non-blocking) |
Show product |
1 mg 10 mg |
mcd25c2-mab10-1
|
Recombinant murinized CD25 antibodies for in vivo use
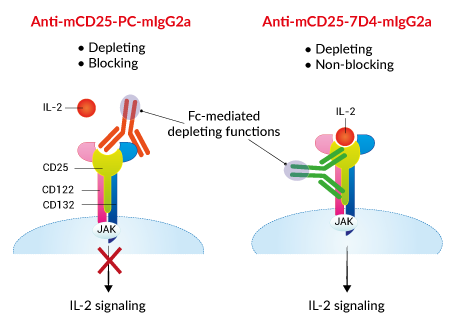
Functions of InvivoGen's Anti-mCD25-mIgG2a antibodies
InvivoGen provides two monoclonal antibodies (mAbs) that target the mouse IL-2R alpha chain (mCD25). They feature a murinized IgG2a constant region and the variable region of either "blocking" or "non-blocking" clones.
— Anti-mCD25-PC-mIgG2a derived from the previously described rat IgG1 clone PC-61.5.3, a "blocking" anti-mCD25 [1,2]
— Anti-mCD25-7D4-mIgG2a derived from the previously described rat IgM clone 7D4, a "non-blocking" anti-mCD25 [2,3]
CD25, in association with CD122 and CD132, forms the high-affinity cell-surface receptor for the IL-2 cytokine (IL-2R). CD25 is constitutively expressed at high levels by regulatory T (Treg) cells and only transiently at low levels by effector T (Teff) cells upon activation [4]. While Teff cell function is augmented by IL-2 signaling, Treg cells absolutely require this signal for survival.
Using recombinant technology, the Fc region of InvivoGen's Anti-mCD25 antibodies has been replaced by a mouse IgG2a isotype in order to mediate strong intra-tumoral Treg depletion. Their variable regions remain different, recognizing distinct epitopes on the CD25 molecule, blocking or not the IL-2 binding.
• The original rat IgG1 PC-61.5.3 mAb mediates in vivo Treg depletion through a single activatory FcγR, FcγRIII, on phagocytes [1,5]. However, this function is hampered in a murine tumor microenvironment due to the local upregulation of the inhibitory FcγRIIb, displaying a similar affinity for rat IgG1 [5]. PC-61.5.3, when turned into mIgG2a mAb, mediates superior intra-tumoral Treg depletion as it binds multiple activatory FcγR (i.e. FcγRI, FcγRIII, and FcγRIV) [5]. The variable region of PC-61.5.3 blocks IL-2 binding to CD25 and thus inhibits IL-2 signaling [2,6].
• The original rat IgM 7D4 mAb is a non-depleting antibody [3]. 7D4, when turned into mIgG2a mAb mediates in vivo depletion of Tregs, in an equivalent manner to PC-derived mIgG2a [6]. The variable region of 7D4 does not block IL-2 binding to CD25 and thus preserves IL-2 signaling [2,6]. This particular aspect has been highlighted as an important parameter to achieve strong antitumor responses in mouse models. Indeed, the data suggest that IL-2 availability after Treg depletion supports anti-tumor Teff cells [6].
InvivoGen's Anti-mCD25 mIgG2a antibodies can be used for in vivo evaluation of anti-tumor monotherapy or combination therapies (i.e. with immune-checkpoint antibodies, or dendritic-cell-based vaccines) in mouse models. Anti-mCD25-mIgG2a mAbs are provided in an InvivoFit™ grade, a high-quality standard specifically adapted to in vivo studies.
Key features:
- Anti-mCD25-PC-mIgG2a is a depleting and blocking antibody
- Anti-mCD25-7D4-mIgG2a is a depleting and non-blocking antibody
- Filter-sterilized (0.2 µm), endotoxin level < 1 EU/mg
- Suitable for parental delivery in mice (azide-free)
- Low aggregation < 5%
-
Produced in animal-free facilities and defined media
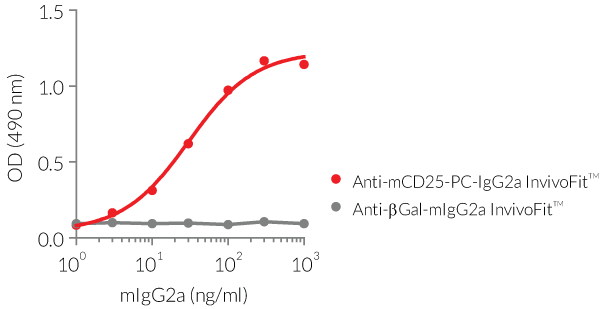
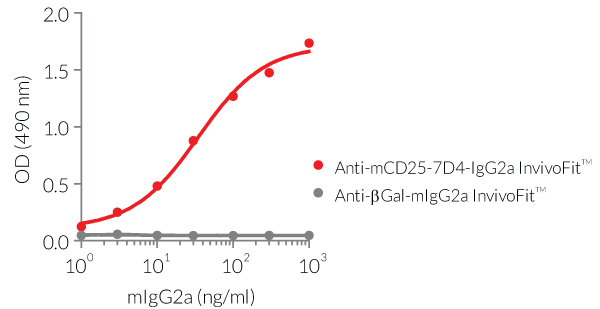
Anti-mCD25-mIgG2a mAbs are produced in Chinese hamster ovary (CHO) cells, purified by affinity chromatography with protein A, and their specific binding to mCD25 has been validated by ELISA (see Figures).
References:
1. Setiady Y. et al., 2009. In vivo depletion of CD4+FOXP3+ Treg cells by the PC61 anti-CD25 monoclonal antibody is mediated by FcgammaRIII+ phagocytes. Eur J Immunol. 40(3):780-786.
2. Moreau J.L et al., 1987. Monoclonal antibodies identify three epitope clusters on the mouse p55 subunit of the interleukin 2 receptor: relationship to the interleukin 2-binding site. Eur. J. Immunol. 17(7):929-935.
3. Malek T.R.. et al., 1983. Identification and initial characterization of a rat monoclonal antibody reactive with the murine interleukin 2 receptor-ligand complex. PNAS 80(18):5694.
4. Damoiseaux J., 2020. The IL-2 – IL-2 receptor pathway in health and disease: The role of the soluble IL-2 receptor. Clinical Immunol. 218:108515.
5. Arce Vargas F. et al., 2017. Fc-optimized anti-CD25 depletes tumor-infiltrating regulatory T cells and synergizes with PD-1 blockade to eradicate established tumors. Immunity. 46(4):577-586.
6. Solomon I. et al., 2020. CD25-Treg-depleting antibodies preserving IL-2 signaling on effector T cells enhance effector activation and antitumor immunity. Nat. Cancer 1(12):1153.
7. Ellis G.L & Riley J.L, 2020. How to kill Treg cells for immunotherapy. Nat. Cancer. 1(12):1134-1135.
Specifications
Specificity: Target cells expressing murine CD25
Formulation: Lyophilized from 0.2 μm filtered solution in 150 mM sodium chloride, 20 mM sodium phosphate buffer with 5% saccharose
Clonality: Monoclonal antibody
Isotype: Murine IgG2a, Kappa
Control: mIgG2a InvivoFit™ isotype control
Source: CHO cells
Purity: Purified by affinity chromatography with protein A
Applications: Flow cytometry; in vivo depletion; ELISA
Quality control:
- The binding of Anti-mCD25-PC-mIgG2a InvivoFit™ and Anti-mCD25-7D4-mIgG2a InvivoFit™ to mCD25 has been confirmed by ELISA
- The complete sequence of antibody constructs has been verified
- < 5% aggregates (confirmed by size exclusion chromatography)
- Endotoxin level <1 EU/mg (determined by the LAL assay)
Contents
Note: these two antibodies are sold separately.
Anti-mCD25-PC-mIgG2a InvivoFit™ and Anti-mCD25-7D4-mIgG2a InvivoFit™ are provided sterile, endotoxin-free, azide-free, and lyophilized.
Each product is available in two pack sizes:
Anti-mCD25-PC-mIgG2a InvivoFit™
- mcd25c1-mab10-1: 1 mg
- mcd25c1-mab10-10: 10 mg
Anti-mCD25-7D4-mIgG2a InvivoFit™
- mcd25c2-mab10-1: 1 mg
-
mcd25c2-mab10-10: 10 mg
![]() The product is shipped at room temperature.
The product is shipped at room temperature.
![]() Store lyophilized antibody at -20 °C.
Store lyophilized antibody at -20 °C.
![]() Lyophilized product is stable for at least 1 year
Lyophilized product is stable for at least 1 year
![]() Avoid repeated freeze-thaw cycles.
Avoid repeated freeze-thaw cycles.
InvivoFit™
InvivoFit™ is a high-quality standard specifically adapted for in vivo studies. InvivoFit™ products are filter-sterilized (0.2 µm) and filled under strict aseptic conditions in a clean room. The level of bacterial contaminants (endotoxins and lipoproteins) in each lot is verified using a LAL assay and a TLR2 and TLR4 reporter assay.
Back to the topDetails
CD25 Background:
The cluster of differentiation 25 (CD25, aka IL-2Rα) is a component of the cell surface receptor for the IL-2 cytokine (IL-2R). It is constitutively expressed at high levels by regulatory T (Treg) cells and only transiently at low levels by effector T (Teff) cells upon activation. CD25 is also expressed on mature dendritic cells.
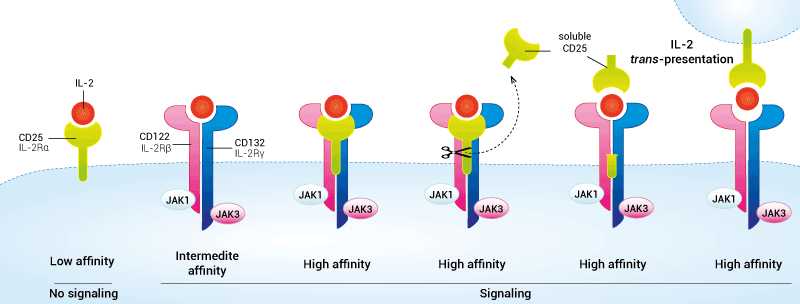
- Role of CD25 in IL-2 cytokine signaling
CD25 exhibits only a low affinity for IL-2 and does not trigger intracellular signaling. The functional IL-2R comprises either 2 or 3 protein chains. CD25 association with CD122 (IL-2Rβ) and CD132 (IL-2Rγ) forms the trimeric high-affinity IL-2R. The association of CD122 and CD132 forms the dimeric intermediate-affinity IL-2R [1]. While Teff cell function is augmented by IL-2 signaling, Treg cells absolutely require this signal for survival.
- Therapeutic targeting of CD25
CD25 has been investigated as a potential target in pre-clinical and clinical tests. Daclizumab, an anti-CD25 commercially available until 2018 was used for the treatment of relapsing forms of multiple sclerosis and the prevention of renal transplant rejection. Its use was based on its ability to block IL-2 binding to CD25 and thus, subsequent IL-2-mediated sustained activation of Teff cells. However, Daclizumab also reduces the systemic abundance of Treg cells in patients, leading to inflammation disorders. Therefore, the repurposing of Daclizumab to preferentially target Treg cells and support anti-tumor responses has been investigated, but poor clinical responses have been reported [2]. A recent report from Quezada's laboratory suggests that the concomitant anti-CD25-mediated blocking of IL-2 sensing by Teff cells prevents efficient tumor rejection [3]. This has led to the engineering of RG6292, an anti-human CD25 that allows Treg depletion while preserving IL-2 signaling and cytotoxic Teff cytotoxic activity [3]. RG6292 is currently being assessed in clinical trials in patients with progressive solid tumors (NCT04158583).
1. Damoiseaux J., 2020. The IL-2 – IL-2 receptor pathway in health and disease: The role of the soluble IL-2 receptor. Clinical Immunol. 218:108515.
2. Bayati F. et al., 2021. The Therapeutic Potential of Regulatory T Cells: Challenges and Opportunities. Frontiers in Immunol. 11:3455.
3. Solomon I. et al., 2020. CD25-Treg-depleting antibodies preserving IL-2 signaling on effector T cells enhance effector activation and antitumor immunity. Nat. Cancer 1(12):1153.